Conversão da Biomassa
Click here to load reader
-
Upload
ctbe-brazilian-bioethanol-scitech-laboratory -
Category
Business
-
view
889 -
download
1
description
Transcript of Conversão da Biomassa

UNIVERSIDADE FEDERAL DE PERNAMBUCODEPARTAMENTO DE ENGENHARIA QUÍMICALABORATÓRIO DE PROCESSOS CATALÍTICOS
RECIFE, PERNAMBUCO
CONVERSÃO DA BIOMASSACesar A. M. Abreu

CONVERSÃO DA BIOMASSA
CONVERSÃO DA BIOMASSA COM VALORIZAÇÃO
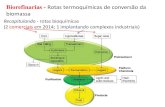
Conversão da biomassaProcessos de conversãoNatureza químicaFracionamentoFuncionalização ou degradaçãoIntermediáriosProdutos finais

CONVERSÃO DA BIOMASSA
BIOMASSA LIGNOCELULÓSICA
Principais componentes: celulose, hemicelulose, ligninaOutros componentes: cinzas, fenois , acidos graxos, ….
Celulose: polissacarídeo de D-glucose, unidades associadas via β-1,4-glucosidic ligações.Hemicelulose: polissacarídeo de xilose, arabinose, manose, promovendo interações entre a celulose e a ligninaLignina: polímero baseado em fenilpropano, estruturado em grupos guaiacil, siringil and p-hidroxifenylpropano

CONVERSÃO DA BIOMASSA
CONVERSÃO DA BIOMASSA (CANA-DE-AÇÚCAR)
1 EXTRAÇÃO SACAROSE
(MELAÇO)
BIOMASSA
L-CEL
(BAGAÇO)
QUÍMICO
BIOQUÍMICO
PRÉ-TRAT.
FRACIONAMENTO
GLUCOSE,FRUTOSE
AÇÚCAR INVERTIDO
CELULOSE
HEMICELULOSE
LIGNINA
2 HIDRÓLISE CELULOSEHEMICELULOSE
ÁCIDO DILUÍDO
ÁCIDO CATALÍTICO
ENZIMÁTICO
GLUCOSE
HMF, DMF
XILOSE, ARABINOSE,
FURFURAL,

CONVERSÃO DA BIOMASSA
CONVERSÃO DA BIOMASSA (CANA-DE-AÇÚCAR)
3 OXIDAÇÃO LIGNINA QUÍMICO
CATALÍTICO
ALDEÍDOS AROMÁTICOS
ÁCIDOS DERIVADOS
4 HIDROGENAÇÃO
HIDROGENÓLISE
OXIDAÇÃO
ESTERIFICAÇÃO
SACAROSE
GLUCOSE
FRUTOSE
XILOSE
(MELAÇO, HIDROLISADOS)
QUÍMICO
CATALÍTICO
POLIÓIS
ÁCIDOS DERIVADOS
ÉSTERES

CONVERSÃO DA BIOMASSA
CONVERSÃO DO BAGAÇO DE CANA-DE-AÇÚCAR
ACETATODE
CELULOSE
SORBITOL /MANITOL
CELULOSE
FURFURAL XILITOL
HEMICELULOSE
RESINASFENÓLICAS
PLÁSTICOS VANILINA
LIGNINA
BAGAÇO DE
CANA-DE-AÇÚCAR

The acid hydrolysis processDilute acid hydrolysis, Low acid consumptionMaximum monosaccharide yields reached at high temperatures and short residence times, Fast reaction ratesYields circa of 50-60% of the theoretical valueConcentrated acid hydrolysis,Processed decomposing and dissolving the polysaccharidesOccurs with water deficiencyProduction of oligosaccharides

The acid hydrolysis processLimitations,Severe conditions (e.g. higher temperature, low pH) Formations of degradation by-products Furans and organic acids Monomeric hexoses and pentoses transformed into HMF and furfural,Further degradation into organic acids (e.g. levulinic, humic acids) and condensation reactions Dissolved lignin result in the formation of inhibiting phenolic compounds Corrosion of the equipment

The acid hydrolysis processProduction process of saccharidic mixtures to further processing,Degradation of corn starch or sugarcane hemicellulose in acid mediaQuantification of the oligomeric decompositions Selection of saccharidic mixtures to further catalytic treatementsKinetics of starch and pentosane depolymerization Consecutive evolutions of the oligomeric components Identification by the degree of polymerizations (DP6, DP5, DP4, DP3, DP2, DP1 = glucose, xylose,..).

The acid hydrolysis processStarch and sugar cane bagasse hydrolysis,Native corn starch solutions were hydrolyzed at temperatures ranging 343 K to 373 K, producing glucose with yield circa 70% Sugar cane bagasse was hydrolised at 393 K, producing xylose, with approximate yield of 60%
Abreu, C. A. M. et al. (1995) Biomass and Bioenergy Vol 9, No. 6, 487-492

The acid hydrolysis processMechanism Kinetics
H -Ac AcH
G G n OHHDP
Ac HDPn AcH DP
- ---------------------------GDP2OHHDP
AcHDP2 AcH DP
GDP1OHSH
Ac SH1 AcH S
'
21
-12
1n
'
2n
nn
n
'
2
-
++⇔
+→+
+→+
+→+
+→+
+→+
+→+
−
−
( )
−−−= GS
'
S'S C(C
kk1Ck
dtdC
O
( ) ( )GSo
1/2
GSo'AcH'G CCCC
kk1Kk
dtdC
−
−−=
( ) ( )
−−−= GSo'S
O CCkk1Ck
dtdC L
GC

CONVERSION OF CARBOHYDRATESProcessing of raw materials rich in saccharides (sugarcane, starch, molasse, bagasse,…), Products with industrial application as polyols and organic acidsCarbohydrate hydrogenations (saccharides → monosaccharides → polyols)
Carbohydrate oxidations (saccharides → monosaccharides + acids → acids)
Heterogeneous processes with supported catalysts based on nickel, chromium, ruthenium to hydrogenate glucose, fructose and sucrose to sorbitol and mannitol

Hydrogenation of carbohydratesHeterogeneous mechanism

1st Brazilian Workshop on Green Chemistry
Hydrogenation of carbohydratesHeterogeneous mechanism

Hydrogenation of carbohydratesSaccharide hydrogenation process,Polyol production in a batch three-phase reactor Glucose conversions of 85% with a selectivity in sorbitol of 99.05% at 413K, under 24 bar, after 3 hours of reaction with a nickel catalyst (14.75 % weight)/activated carbon Saccharose conversions of 52% after 3 hours of reaction Production of glucose and fructose and sorbitol and mannitol
L. C. A. Maranhão, F. G. Sales, J. A. F. R. Pereira, C. A. M. Abreu (2004) React. Kinet. Catal. Lett. 81, 169-175

Hydrogenolysis of carbohydrates
Saccharide hydrogenolysis process,More drastic temperature and hydrogen pressure conditionsSplitting of carbon-carbon and carbon-oxygen carbohydrate bonds Polyols obtained from hydrogenations can be hydrogenolysedProducts: other polyols, glycols and alcoholsCatalysts: noble metals

Continuous production of fine polyols
Scale-up of carbohydrate hydrogenations,Fine polyols from biomass resources are traditionally produced in discontinuous processesApparatus of great volume in relation to the small quantity of the obtained productsScale-up from discontinuous operations to continuous oneDevelopment of the saccharide hydrogenation process into a continuous operationContinuous polyol production

Continuous production of fine polyols
Continuous hydrogenation in a three-phase reactor,Trickle-bed reactor under moderate operation conditions (1.22 MPa, 413 K)Glucose conversions of 44% with a polyol selectivity of 99.31%Yield of 24% in sorbitol and mannitol for the saccharose hydrogenation Possibility to develop a process (pressures up to 2.54MPa, low liquid flow rates) to obtain high conversions
Maranhão, L. A., Abreu, C. A. M. (2005) Industrial and Engineering Chemistry Research. v. 44, p. 9642-9645

Continuous production of fine polyols
0,0 0,1 0,2 0,3 0,4 0,5 0,6
0,0
0,1
0,2
0,3
0,4
0,5
glucose sorbitol model
C (m
ol L-1 )
Axial position (m)
Hydrogenation of glucose at 1.22MPa and 413K in trickle-bed reactor
012 =+
′−−
GG
GGGGL
Gax CK
Ckdz
dCu
dzdC
Dη
( ) ( )[ ]( ) ( ) ( )[ ]GeLG
GeGeG fSh
ff
Gφφφ
φφφη
3f3coth13f3coth
eG
eG
−+−
=

Continuous production of fine polyols
0,0 0,1 0,2 0,3 0,4 0,5 0,6
0,0
0,1
0,2
0,3
saccharose monosaccharides polyols modelC
(mol
L-1)
Axial position (m)
Hydrogenation of saccharose at 1.22MPa and 413K in trickle-bed reactor.
02
2
=′−− SacSacSacSac
LSac
ax Ckdz
dCu
dzCd
D η
012
2
=
+′
−′+−MoMo
MoMoSacSacMo
MoMoax CK
CkCk
dzdC
udzCd
D η
012
2
=+′
+−MoMo
MoMoPo
PoPoax CK
Ckdz
dCu
dzCd
D η

Continuous production of fine polyols
An up grade of the discontinuous to the continuous process for saccharide hydrogenation may be compared in the following terms: Discontinuous process (slurry reactor) Continuous process (trickle-bed
reactor) Ni/C catalyst; 413 K, 2.44 MPa Ni/C catalyst; 413 K, 1.22 MPa Operation time = 3 hours Operation time = 3 hours Concentration of the saccharide feed = 100.00 g/L
Concentration of the saccharide feed = 100.00 g/L
Production = 42.50 g in polyol Acumulated production = 749.35 g in polyol

LIGNIN FROM BIOMASS
Biomass conversion into aldehydes and acids,Lignin degradation: break up into fragments producing aromatic aldehydesPolifenate ions, precursors of the aromatic aldehyde formationsAldehyde conversion into organic acids

LIGNIN PROCESSING FROM SUGARCANE BAGASSE
Lignin oxidation,Wet air oxidation process (WAO) as an alternative technology Valorization of lignocellulosic materialsProduction of a mixture of aromatic aldehydes of industrial interestCatalytic wet air oxidation (CWAO) process using air and catalysts Treatment of effluents and by-product of the biomass industry

Catalytic wet oxidation of lignin
→ − 322 OAl / γPd / O → − 322 OAl / γPd / O → − 322 OAl / γPd / O2
OH
C
R2R1
HO O
CO
H3COO
C
O
CH
OCH3H3CO
OCH3
OCH3
O
C
C
OOCH3
CH
H2COH
H2COH
H
HC
CHO
H2COH
H
H2COH
H1
2
3
4
(a)
H2COH
H
2
C
O
CH
OCH3H3CO
C
OH
C
R2R1
OHHO H
H
2
H2COH
[ Lignin ] [ Aldehydes ] [ Acids ]
(b)
2
OH
C
R2R1
OH
+ AcH + AcH
Basic structure of lignin and degradation/oxidation mechanism. (a) basic unit of the Fagus silvatic lignin. (b) degradation/oxidation reaction steps. R1= H, OCH3 ; R2 = OCH3 .

Catalytic wet oxidation of lignin
Reaction scheme of the catalytic wet oxidation of lignin
CWAO of lignin from sugar-cane bagasse was evaluated to produce aromatic aldehydesLignin (L) is depolymerized with the productions of aldehydes, acids and other products of low molecular weights The aromatic aldehydes vanillin (V), syringaldehyde (S) and p-hydroxibenzaldehyde (P) were submitted to subsequent oxidationsOther products (R), such as organic acids can degrade into carbon dioxide

Operations in a slurry reactor,Palladium catalyst, 373-413 K, 2-10 bar/ PO2 Lignin as a by-product from sugarcane bagasse by the DFH (Dedine Fast Hydrolysis) Yields of the aromatic aldehydes associated with lignin consumption and their oxidations to acidsAromatic aldehyde yields approximately ten to twenty times higher then with the noncatalytic oxidation process
Sales, F. G. , Maranhão, L. A. , Lima Filho, N. M. , Abreu, C. A. M.( 2006). Industrial & Engineering Chemistry Research. v. 45, p. 6627-6631
Process operations

Processo continuo de produção de aldeídosaromáticos
Scale-up of process,From batch to continuous operations
Aromatic aldehyde productions operated in a continuous fluidized-bed reactor
Lignin as a by-product from sugarcane bagasse
Yields of the aromatic aldehydes associated with the lignin consumption and their oxidations to acids

Processo continuo de produção de aldeídosaromáticos
Three-phase fluidized-bed reactor

Processo continuo de produção de aldeídosaromáticos
Escalonamento,
Batch operation: 56.24x10-2g of vanillin and 50.01x10-2g of syringaldehyde from a 0.50L-lignin solution (60.00g/L), 2 h of reaction at 5.00 bar and 393 KContinuous operation: 65.10x10-1g of vanillin and 114.84x10-1g of syringaldehyde, with a feed concentration of lignin of 30.00 g/L, 2 h of reaction, at 5.00 bar and 393 K, liquid-phase flow rate of 5.00 L/h
F. G. Sales, L. C.A. Maranhão, N. M. Lima Filho, C. A.M. Abreu (2007) Chemical Engineering Science 62, 5386 – 5391

Recent technology developments done in the scope of the biorefinery concept have emerged as alternatives, making production of chemicals from ligno-cellulosic feedstocks become a reality.
Biomass conversions employ hydrolysis and pretreatments of hemicellulose and lignin, and acid or enzymatic hydrolysis of cellulose to break the polymeric structures to their saccharides and lignin components.
In the presence of homogeneous or heterogeneous catalysts the oligomeric mixtures selected may be processed in order to produce valuable chemicals.
Through catalytic hydrogenation, hydrogenolysis or oxidation these mixtures can be converted to polyols, glycols, monoalcohols, aldehydes and organic acids.
Conclusions